
Institute
of Optoelectronics, M.U.T., 2
Kaliski Str., Warsaw, 01-489, Poland
*
Institute of Physics, P. A. S., 32/46 Al. Lotników, 02-668 Warsaw, Poland
**
Institute of Electronic Materials Technology, 133 Wólczyńska Str., Warsaw,
01-919, Poland
Abstract.
Growth and recharging processes of chromium ions in SrGdGa3O7
and SrLaGa3O7 single crystals incorporated during growth
or doping by diffusion (only for SrLaGa3O7 crystal) were
analyzed. The annealing at 1000oC in air and ionizing with gamma rays
(doses from 103 to 105 Gy) and protons (1014 cm-2
only for SrGdGa3O7 crystal) were performed. Different
kinds of chromium ions valency, from two to four, were found. Electron spin
resonance measurements in the temperature range 5-300K with the use of Brucker
spectrometer were also done.
Keywords:
crystal growth, additional absorption, gamma exposure, proton irradiation,
electron spin resonance
Symbole
UKD: 548.73
Cr3+:SrGdGa3O7
crystals (Cr:SGGO) and Cr:SrLaGa3O7 crystals (Cr:SLGO)
belongs to the family of alkaline and rare-earth gallates, which are described
by general formula ABC3O7. They have the tetragonal
gehlenite (Ca2Al2SiO7) structure (space group
P421m., D32d). Unit cell parameters of SLGO and
SGGO are: a=0.8058 nm, c=0.5333 nm and a=0.79868 nm, c=0.5255 nm, respectively. These
crystals can be doped by any of rare-earth elements (Ce to Eu instead of La and
Tb to Lu instead of Gd). In addition, a part of Ga ions can be replaced by Cr
ions. Bivalent Sr ions and trivalent rare-earth ions occupy equivalent
crystallographic positions with stochastic distribution, therefore these
materials exhibit some glass-like properties. However, they show high thermal
conductivity and other features characteristic for ordered crystals. Thus, this
class of materials is very interesting for both basic research and laser
technology.
Some
of the members of the family of compounds Nd3+: BaLaGa3O7
(Nd: BLGO) and Nd3+: SrLaGa3O7 (Nd: SLGO) were
investigated elsewhere [1-3]. Measurements of spectral and lasing
characteristics of Nd: SGGO crystals doped with 1.2 at. % of Nd3+ and
Cr: SLGO crystals doped with 0.1 at. % of Cr3+ were presented in
[4-6]. Spectroscopic characteristics (absorption at 300K and 77K) of Cr: SGGO
and Cr: SLGO single crystals has been presented in [7]. It was found, that the
color of the crystals is associated mainly with the absorption of Cr4+
ions that substitutes Ga3+ ions and structural defects resulting from
stoichiometry deviations.
In the
present paper changes in the valency of chromium ions in Cr: SLGO and Cr: SGGO
single crystals and Cr: SLGO doped by diffusion under ionizing radiation and
thermal annealing treatments are investigated.
Chromium
doped SLGO and SGGO single crystals have been grown by the Czochralski method in
an atmosphere of nitrogen with an admixture of 1% oxygen. Starting
concentrations of Cr in the melt were 0.05, 0.1 and 0.25 at. % with respect to
Ga. Crystals with 18 mm diameter were pulled from a 40 mm diameter iridium
crucible on <001> oriented seeds. The pulling rate ranged from 1.5 to 4
mm/h. Convex crystal - melt interface was kept during the whole crystal growth
process.
Chemical
composition of both crystals slightly differs (0.7 - 1.2 at. %) from
stoichiometric one and therefore, they may be treated as solid solutions of
gehlenites and SrGa2O4. Uniform distribution of Sr, La/Gd
and Ga along crystal growth direction has been confirmed by Electron Probe
Microanalysis method. This fact implies that the starting melt composition was
properly chosen and did not change during the crystal growth process. Chromium
doped crystals are diachronic with blue color. The color intensity is correlated
with Cr concentration.
Fig. 1
present photos of the two investigated crystals with Cr concentration equal to
0.25 at. %. It can be seen, that Cr: SLGO crystal show non-homogeneity in Cr
distribution along the crystal. Homogeneous Cr distribution was obtained for all
SGGO crystals but in the SLGO only for 0.05 and 0.1 at. % Cr concentrations.

Fig. 1. Cr:SrLaGa3O7
single crystals as compared to Cr:SrGdGa3O7. Concentration
of chromium ions for both crystals was equal to 0.25 at. %.
SLGO plates embedded in the powdered Cr2O3 were heated in air at two temperatures: at 900oC for 20h and at 1000oC for 30h. After each diffusion process plates were irradiated by gamma’s with a dose of 105 Gy.

Fig. 2. ESR signal from Cr: SGGO crystal at 5K for two orientation of
magnetic field with respect to c-axis: 90o and 0o.
Both
sides polished samples 1-2 mm thick were optically investigated before and after
the next treatments: g-irradiation
with doses 103-105 Gy from 60Co source, proton
irradiation’s with an energy of 21 MeV and a fluency of 1014 cm-2
from C-30 cyclotron and thermal annealing at 1000oC in air for 3h.
Eight samples of SGGO crystal were investigated: SRGD1...SRGD6, SGGOCR1 and
SGGOCR2 and two of SLGO. The absorption spectra at 300K and 77K were measured
using LAMBDA-2 Perkin-Elmer spectrometer. Additional absorption (AA) bands
induced by gamma’s, protons and annealing were defined as follows:
![]() (1)
(1)
where:
d is the thickness of the sample, T1, T2 are the
transmissions of the sample before and after irradiation or annealing,
respectively.
Electron
spin resonance (ESR) spectra were recorded at 9.4 GHz, at temperature range from
4 K to 300 using BRUKER ESP -300 X-band spectrometer.
Fig.
2 presents ESR spectra for two positions of magnetic field vector with respect
to c-axis of the Cr: SGGO crystal for microwave power P=0.2 mW and at a
temperature of T=5 K. The width of the ESR spectra for Gd is about 450 mT and
therefore chromium lines are not observed. Fig. 3 shows change in ESR signal
amplitude v.s. temperature, from 5K to 300K. No lattice relaxation and pair of
ions interaction is observed. For so great concentration of Gd3+ ions,
by our opinion, the strong interaction between pairs of Gd3+ ions
should exist.
In Fig. 4a absorption spectra for six investigated crystal samples are depicted. Fundamental optical absorption edge (FAE) begins at 250 nm and shifts towards the longwave direction by about 10 nm with an increase of Cr3+ concentration from 0.05 to 0.1 at. %. Clear bands can be distinguished, at: 253, 274, 306, 311, 450, 548, 600, 706 and 775 nm in UV and VIS region of absorption spectrum. The bands with maxima: 274, 306 and 311 are connected with Gd3+ ion in position of Ga3+. Bands with maxima at: 548 and 600 nm one can be treated as transition inside Cr3+ ions, while 706 and 775 nm bands as transitions inside Cr4+ ions. The band at 253 nm is probably associated with non-controlled ions, e.g. Fe3+, coming from Cr2O3 oxide, while the band at 450 nm with Cr2+ ions. The above assumption is confirmed by irradiation and annealing treatments of the investigated crystals, according to results presented in Figs 4b, c. After g-irradiation at 300 K at least three AA bands can be distinguished.
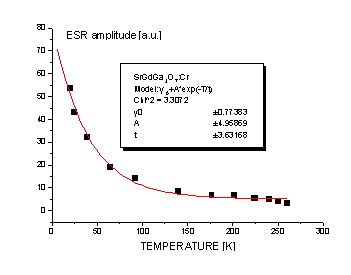
Fig. 3. Temperature dependence of ESR signal for Cr: SGGO crystal in the range
of 5-300 K
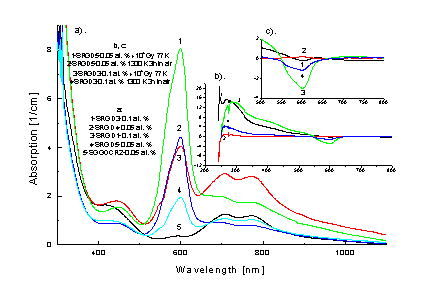
Fig. 4. Absorption (a)
and AA (b, c) in Cr: SGGO single crystal after
g-irradiation with doses 103-105 Gy
The
first one (about 16 cm-1 in intensity), in dependence on Cr
concentration, is centered near 270 nm or 290 nm centered, for 0.05 and 0.1 at.
%, respectively. This band is probably associated with Ga3+®
Ga2+ reaction, which leads to paramagnetic defect of G1 type,
observed earlier for SLGO single
crystal in [3].
Second
band, at 364 nm, in our opinion, is associated with oxide vacancies. Relative
intensity of the two maxima clearly depends on dose value. With an increase of a
dose from 103 to 105 Gy, intensity of the first maximum
increases while that of the second diminishes. Similar results for Nd: SLGO
single crystal were earlier obtained [3].
Third
band, at 600 nm, with values of AA <0 correspond to a decrease in Cr3+
concentration.
Moreover,
the wide AA bands with maxima at 706 and 775 nm are observed. They can be
connected with a change in Cr4+ concentration. The g-irradiation,
performed at 77K does not introduce additional AA bands. Also an increase in AA
intensity with respect to g-irradiation
at 300K is not observed.
Accordingly
to Fig. 4b, c annealing of Cr: SGGO crystals in air for 3h at 1000oC
gives at least two AA bands, first at 270 nm and second at 600 nm. The second
band can be connected with a change in Cr3+ concentration and
effectively in Cr valency. For all the treatments changes near 450 nm band is
also seen.
Among
six investigated samples there are three the most characteristic: SRGD3 (0.1 at.
% Cr; 8 cm-1 at 600 nm), SRGD6 (0.05 at. % Cr; 4 cm-1 at
600 nm) and SRGD5 (<0.05 at. % Cr; 1.8 cm-1 at 600 nm). For SGDR3
specimen a decrease in Cr3+ concentration after both annealing and
irradiation is observed which suggests that the main mechanism of valency change
in chromium is ionization process of
the type: Cr3+®
Cr4+. So, in SRGD3 sample Cr3+ and Cr4+ ions
may be present. For SRGD6 sample Cr3+ concentration increases after g-irradiation which means that Cr4+®Cr3+
reaction takes place, probably due to Compton effect. So, in SRGD6 sample Cr3+
and Cr4+ ions can be also present. For SRGD5 crystal an increase in
Cr3+ and Cr4+ concentration after annealing process at
1000oC is observed which suggests the presence of Cr2+
ions and Cr3+ ions in the crystals. An ionization with gamma’s
leads to an opposite effect: Cr3+®Cr2+.
So, in SRGD5 sample Cr3+ and Cr2+ ions should be present.
In Fig. 5 changes in absorption spectrum of Cr: SGGO crystal after g-irradiation
with doses of 103 and 105 Gy and protons with fluency of
1014 cm-2 are presented. It can be seen from Fig. 5a that
change in valency of chromium ions depend on value of dose and is greater for
higher dose. After a time interval of 0.5 year from g-irradiation with a dose of
105 Gy, intensity of AA bands is of the same order of magnitude as
intensity of AA immediately after g-irradiation
with a dose of 103 Gy. Moreover, in the range of 600 nm band no
changes are observed, which means that recharging of chromium ions due to
ionization is stable in time.
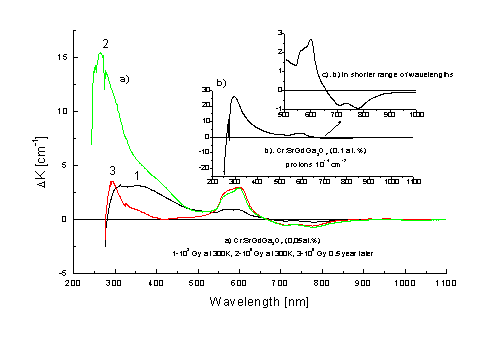
Fig. 5.
AA bands in Cr: SGGO crystal for different doses of g-irradiation
and after relaxation in time for 0.5 year at 300K (a) and protons with a fluency
of 1014 cm-2 and energy of 21 MeV (b, c)
Fig. 5b
show changes in absorption of Cr:SGGO crystal after proton irradiation. The
changes are similar as for g-irradiation.
ESR measurements did not show any Cr lines in these crystals, which was an unexpected effect. High level of defective structure of the crystals can be a possible explanation of this phenomenon.
Fig. 6 illustrates changes in valency of Cr ions in SLGO single crystal doped during growth as compared to SLGO specimens with Cr introduced by diffusion. According to Fig. 6a, during a diffusion process, Cr ions substitute mainly positions that give absorption characteristic for oxide vacancies (AA band at 364 nm). An increase of diffusion temperature leads to an increase in Cr concentration. Changes over 364 nm band after ionizing radiation process with gamma’s are associated mainly with valency changes of Cr ions (also the band at ~280 nm is seen) and means that recharging process of the type: Cr3+®Cr4+ (ionization process) takes place. Excitation of the band with a wavelength of 355 nm gives emission at 458, 487 and 580 nm.
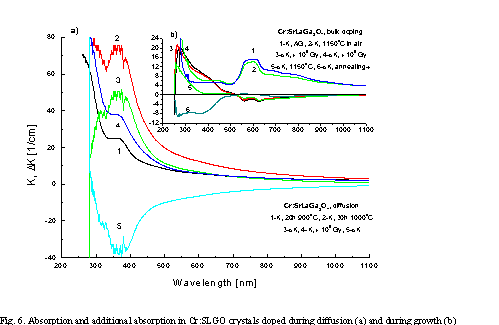
From
Fig. 6b it is evident that for „as grown” Cr: SLGO crystal the same effect
as for diffusive doping is observed, because annealing and irradiation processes
give the same result: a decrease of Cr3+ concentration. Moreover, in
contrary to Cr: SGGO crystal, the band at 364 nm is higher, which means stronger
defective structure of Cr: SLGO lattice as compared to Cr: SGGO crystal (more
oxide vacancies). This observation is in agreement with an earlier
one describing difficulties in obtaining homogeneous distribution of Cr ions in
the case of higher doping (0.25 at. %). Cr: SLGO crystal, for which results of
investigations are presented in Fig. 6b was firstly g-irradiated
with a dose of 105 Gy, then annealed at 400oC, next again
irradiated with the same dose and finally annealed for 17h in air at 1150oC.
Curves 4 and 5 clearly show that the crystal is stable with respect to g-irradiation.
Similarly
as for Cr: SGGO crystals, bands at 548 and 600 nm are electron transition bands
inside Cr3+ ion and bands at 706 and 775 nm are electron transition
bands in Cr4+ ion. So, in „as grown” Cr: SLGO crystals Cr3+
and Cr4+ are simultaneously observed in optical absorption spectrum.
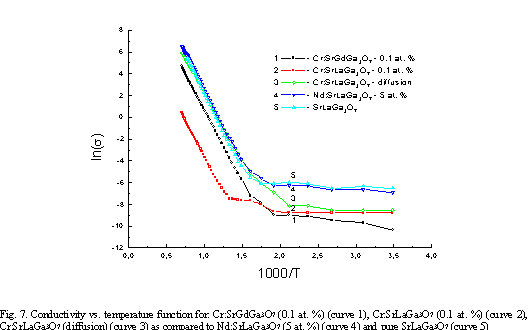
In
Fig. 7 conductivity vs. temperature function is presented for Cr: SGGO, Cr: SLGO,
Cr: SLGO (diffusion) as compared to Nd: SLGO and pure SLGO crystals. The band
gap value calculated from the slopes of the presented curves is equal to about
2.4 eV. It can be seen that more defective structure shows Cr: SLGO
crystal. Moreover, it is seen also that Cr: SGGO crystal exhibit small
dopant conduction.
In
crystals grown at a relatively high pulling rate the fluctuations of chromium
content were visible. To obtain crystals with uniform distribution of Cr it is
necessary to pull them at a sufficiently low rate (below a certain critical
value dependent on Cr content), <1.5 mm/h.
In
the investigated Cr: SGGO crystals the presence of Cr2+, Cr3+
and Cr4+ ions was detected. Their concentration depend on initial
concentration of Cr3+ ions in the melt. Changes in absorption after
annealing or ionizing treatments depends also on this initial concentration. For
greater values of Cr3+ concentration Cr3+®
Cr4+ reaction (ionization) during
annealing and irradiation treatments are preferred. For lower values of Cr3+
concentration Cr2+®Cr3+
reaction during annealing and Cr3+®Cr2+
during irradiation are preferred. So, for greater Cr3+ concentration
Cr3+ and Cr4+ ions are present in Cr: SGGO lattice,
whereas for lower initial concentration of Cr3+ ions, Cr3+
and Cr2+ ions are seen in optical absorption spectrum.
In Cr:
SLGO crystals transitions in Cr3+ and Cr4+ ions are seen
in absorption spectrum. After thermal or ionizing treatments ionization takes
place as a result of reaction: Cr3+®Cr4+.
The same phenomenon is observed for Cr: SLGO crystals doped by diffusion process.
Cr ions locate mainly in lattice sites characteristic for oxide vacancies,
formed in the crystal during growth process.
The
structure of Cr: SLGO crystal is more defected than Cr: SGGO one. Probably, it
is a reason why less Cr ions can be introduced into this crystal.
Absorption
spectrum of Cr: SGGO crystal shows bands characteristic for Cr3+ ions
also as opposite to previous investigations, where Cr4+
bands only were detected [8].
[1] W.
Ryba-Romanowski, M. U. Gutowska, W. Piekarczyk, M. Berkowski, Z. Mazurek and B.
Jeżowska-Trzebiatowska, „Spectroscopic investigations of neodymium-doped
BaLaGa3O7 single crystals”, J. of Luminescence, 36
(1987) 369-372
[2] W.
Ryba-Romanowski, B. Jeżowska-Trzebiatowska, W. Piekarczyk and M. Berkowski, „Optical
properties and lasing of BaLaGa3O7 single crystals doped
with neodymium”, J. Phys. Chem. Solids,
49 (2) (1988) 199-203
[3] S.
M. Kaczmarek, K. Kopczyński, Z. Mierczyk, „Spectroscopic and lasing
properties of SrLaGa3O7:Nd3+”, Proc. of SPIE, vol. 2202 (1995) 182-184
[4] F.
Hanson, D. Dick, H, R. Verdun, M. Kokta, „Optical properties and lasing of
Nd:SrGdGa3O7”, J. Opt. Soc. Am. B/ vol. 8, No. 8 (1991)
1668-1673
[5] K. R.
Hoffman, W. Jia, Phys. Rev.
B 49, 15505 (1993)
[6] S.
G. Demos, V. Petricevic, R. R. Alfano, Phys. Rev. B52, (1995) 544
[7] W.
Ryba-Romanowski, S. Gołąb, G. Dominiak-Dzik, W. A. Pisarski, M. Berkowski and
J. Fink-Finowicki, ”Growth and spectroscopy of chromium doped SrXGa3O7
(X=La, Gd) crystals”, Proc. of Excited
States Conference, Duszniki Zdr., 1997
[8] M.A.
Scott, D.L. Russell, B. Henderson, T.P.J. Han, H.G. Gallagher, Crystal growth
and optical characterization of novel 3d2 ion laser hosts, J. Of Crystal Growth,
183 (1998), 366-376
S.M.
KACZMAREK, M. BERKOWSKI,
R. JABŁOŃSKI
I.
PRACKA, M. KWAŚNY,
M. ŚWIRKOWICZ
Streszczenie.
Analizowano proces wzrostu i procesy zmiany walencyjności jonów chromu w
monokryształach SrGdGa3O7 i SrLaGa3O7.
Chrom wprowadzano podczas procesu wzrostu oraz w wyniku procesu dyfuzji (tylko
dla kryształu SrLaGa3O7). Przeprowadzono wygrzewanie w
temperaturze 1000oC w powietrzu oraz naświetlanie kwantami gamma
(dawki 103-105 Gy) i protonami (1014 cm-2
tylko dla kryształu SrGdGa3O7). Stwierdzono występowanie
jonów chromu o walencyjności od 2 do 4. Przeprowadzono również pomiary
elektronowego rezonansu spinowego w zakresie temperatur 5-300 K z wykorzystaniem
spektrometru firmy Brucker.